Latest from the Alliance
News
Blogs
Publications
Featured
-
Report urges government to back universities’ high-level skills provision
The Social Market Foundation’s ‘Skills Sidelined’ report has called on the government to back universities providing high-level skills provision to plug skills shortages.

-
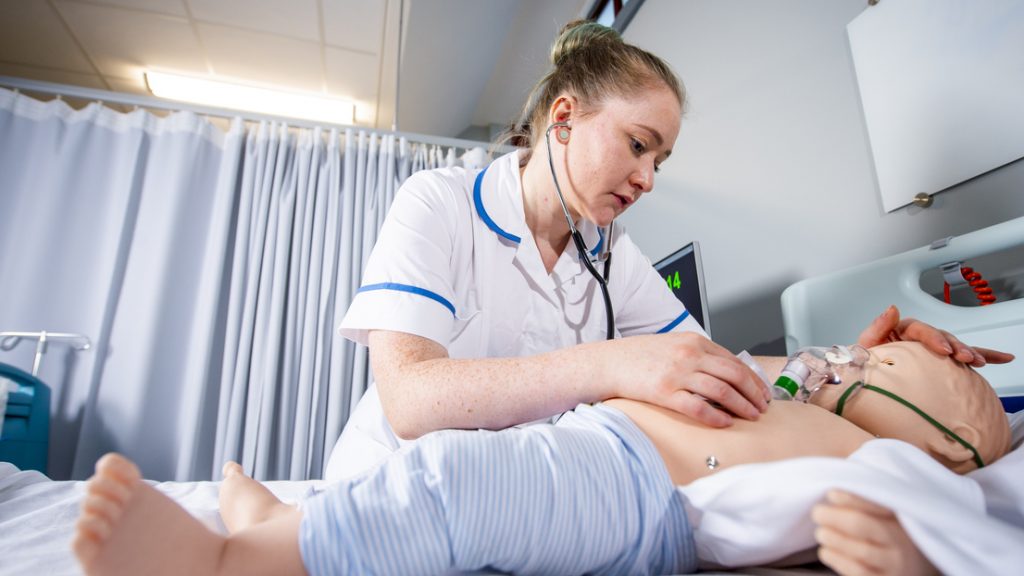
New report on the barriers to expanding healthcare degree apprenticeships
University Alliance (UA) and the University of Derby have jointly published a comprehensive report that explores the current enablers of and barriers to delivering healthcare apprenticeships.

-
Let’s Get Technical: Priorities for the new government
With a change of government taking place In summer 2024, we published an updated policy publication, titled: Let’s Get Technical: Priorities for the new government.

-
To what degree: Understanding what UK businesses look for in graduates
When hiring, employers value a graduate’s skills, the subject they studied, and vocational experience during their degree ahead of grades achieved or whether the graduate studied at a particular university.

UA in facts and stats
95%
…of graduates from Alliance Universities were in work or further study (or another chosen activity) just 15 months after graduation.
93%
…of UA graduates in work (or further study) describe their work as meaningful.
24k
…businesses and organisations are supported to start up and grow every year by an Alliance University.