Featured
-
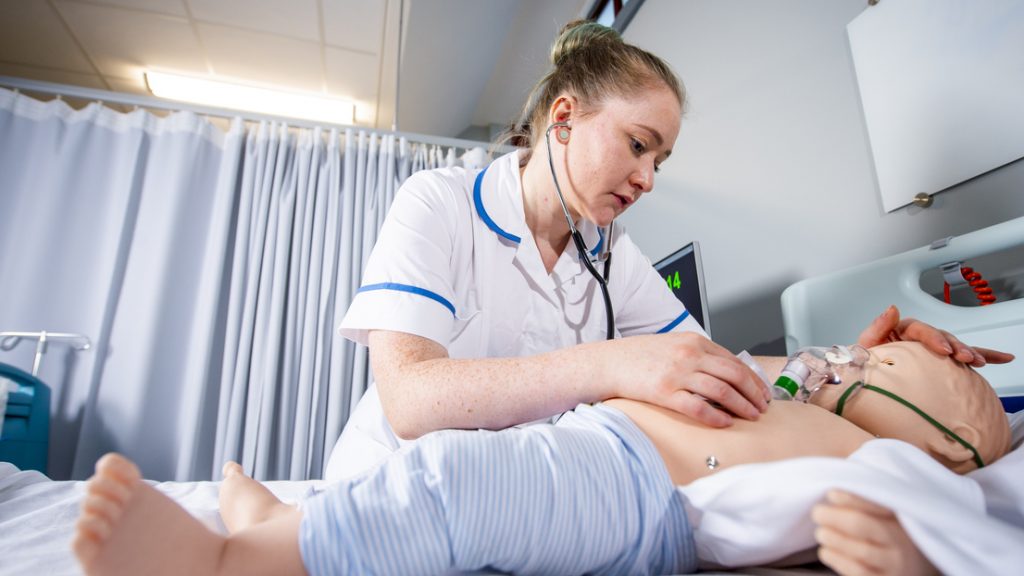
New report on the barriers to expanding healthcare degree apprenticeships
To coincide with National Apprenticeship Week, University Alliance (UA) and the University of Derby have jointly published a comprehensive report that explores the current enablers of and barriers to delivering…

-
Let’s Get Technical: Priorities for the government
In 2023, we published Let’s Get Technical, our manifesto for professional and technical universities. With a change of government taking place In summer 2024, we published an updated policy publication,…

-
To what degree: Understanding what UK businesses look for in graduates
When hiring, employers tend to value a graduate’s skills, the subject they studied, and vocational experience gained during their degree ahead of grades achieved or whether the graduate studied at…

Latest from the Alliance
News
Blogs
UA in facts and stats
95%
…of graduates from Alliance Universities were in work or further study (or another chosen activity) just 15 months after graduation.
93%
…of UA graduates in work (or further study) describe their work as meaningful.
24k
…businesses and organisations are supported to start up and grow every year by an Alliance University.